Quality Management
The company has established a quality assurance system and comprehensive organizational structure to meet the GMP requirement. The person in charge of quality management and the person in charge of production management have distinct and independent responsibilities to the company . The authorized quality person system has been established in the company and every employee from each department has the written job description, and based on it the responsibilities are assigned and implemented. Depending on the production requirements, proper resources such as manpower, plants, facilities and equipment are allocated, together with the quality assurance and quality control system and quality management document system, the company ensures that the quality of the product produced are consistent, safe and effective, and meet the standards for intended use and registration requirements.
stable quality
safe and effective
meet the requirements

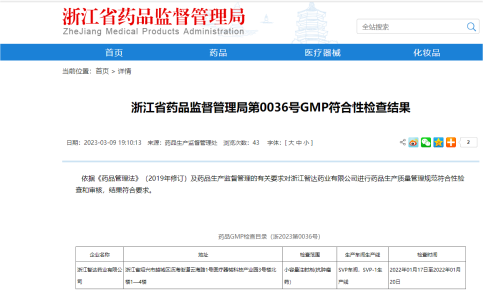
All personnel involved in the production of pharmaceutical products are trained in accordance with GMP requirements and the requirement of internal quality assurance system. “Good Manufacturing Practice for Drugs” and the “People‘s Republic of China Drug Administration Law” are strictly implemented in all aspects of drug production and distribution from raw material procurement, inspection and testing, production to sale and distribution. The company's production lines has passed the GMP compliance inspection.
SFDA
GMP


 R&D system
R&D system Quality Management
Quality Management Technology Platforms
Technology Platforms